MEASUREMENT TOOLS
State-of-the-art measurement systems
Complementary lab solutions
We are official distributors of redox.me novel measurement setups which are ideal for laboratory testing and / or research applications.
These modern electrochemical cells simultaneously allow for the measurement and analysis of various properties, such as optical, thermal, electrochemical and others…
Thin material layers such as for example, coatings or even liquids, can be thoroughly examined by these unique yet complementary lab solutions, now that, several measurement methods are combined.

The broad range of available systems is listed below. In addition, new solutions are currently being developed. Cells can also be customized and tailored to cover any specific application requirements:
- Quartz crystal microbalance electrochemical cells.
- Bottom mount electrochemical & corrosion cells.
- Basic and two-compartment electrochemical cells.
- Photo-electrochemical double & single sided cells.
- Optical fiber spectro-electrochemical flow cell.
- Gas Diffusion Electrode X-Ray diffraction electrochemical flow cell.
- Electrolyte-gated transistor bottom mount electrochemical cell
- Standard, small. large and double-tank etch cells
- Bulk electrolysis basic & two-compartment cells
- Spectro-electrochemical flow cells
- Raman Electrochemical Flow Cell
- Photo-electrochemical H-cells
INNOVATIVE LAB TOOLS
Research Products
Quartz crystal microbalance electrochemical cells
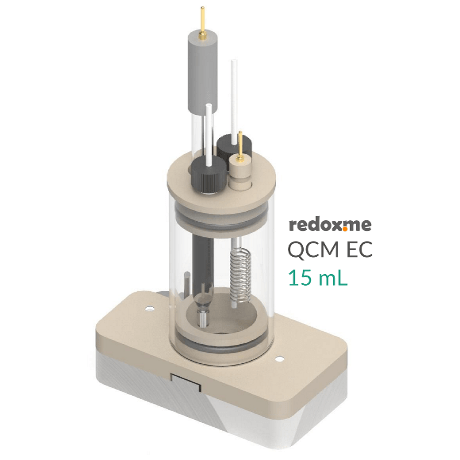
A fixed solution voltammetric cell for the simultaneous measurement of electrochemical responses plus quartz crystal microbalance sensing.
These modules were developed for specific equipment provided by Stanford Research Systems and Biolin Scientific.
Bottom mount electrochemical & corrosion cells

- Thin metal film deposited on flat substrate.
- Metal plate.
- Metal mesh.
- Metal wire.
- Metal ribbon.
Basic & two-compartment electrochemical cells

Fixed solution basic and two-compartment electrochemical cells for measuring electrodes of the following forms:
- Rod / disc (6 mm diameter).
- Thin film deposited on flat substrate (using a wire clip).
- Membrane (using a wire clip).
The structure is gas-tight and can be used by bubbling an inert gas through the electrolyte to remove or exclude contaminants such as oxygen and water.
Photo-electrochemical single & double sided cells

These are horizontally mounted single and double chamber photoelectrochemical cells. These units allow for a variety of methods in order to carry out measurements in 2 or 3 electrode settings.
The structure is gas-tight with gas inlets and outlets that can be used to bubble the solution and vent gases from the chamber.
Optical fiber spectro-electrochemical flow cell

Spectroelectrochemical cells are used to study the relationship between absorbance / transmittance and electrochemical potential, thereby, they can effectively monitor the optical changes of electrochromic materials in real time.
Gas Diffusion Electrode X-Ray diffraction electrochemical flow cell

A GDE XRD electrochemical cell is used to efficiently screen and validate GDEs (Gas Diffusion Electrodes) for metal-air batteries, water electrolysis and alkaline fuel cells.
It also enables acquiring X-ray diffraction measurements in-situ.
It is used as an aspirating, thermodynamic electrical analysis reference unit which simulates the Janus arrangement of a GDE in a metal-air battery: one side is freely exposed to the gas and the other side is wetted by the electrolyte.
Electrolyte-gated transistor bottom mount electrochemical cell

Fixed solution electrochemical cell built to measure the characteristics of an electrolyte-gated (solution-gated) transistor using a liquid or gel electrolyte.
The fabrication of a measured transistor sample can be realised on a rigid or flexible flat substrate (not included in the system) having a conductive thin film source, as well as drain, and a layer of conductive or semi-conductive channel material.
Standard, small, large and double-tank etch cells

For example, a double-tank etching cell is generally used to produce a porous Si thin film by anodization. The process of forming porous Si is performed in a constant manner by applying a current between two platinum electrodes.
Bulk electrolysis basic & two-compartment cells

Standard and two-chamber high-capacity electrolytic cells (also known as constant-potential or controlled-potential coulometric cells) with basic construction.
The structure is gastight.
Spectro-electrochemical flow cells

These are horizontally mounted, multi-purpose spectroelectrochemical cells designed to be utilized with a spectrometer (UV-Vis, or IR). They are capable of obtaining electrochemical and spectral information from a liquid sample or thin-film in systems where a change in the applied potential causes a variation in the observed spectrum.
Spectroelectrochemical cells are used to study the relationship between absorbance/transmittance and electrochemical potential, thereby, they can effectively monitor the optical changes of electrochromic materials in real time.
In-situ spectral acquisition is readily obtained for the detection, identification and characterization of short-lived electrochemically generated species / intermediates, which in turn derives to a detailed investigation of the mechanisms of redox reactions.
Raman Electrochemical Flow Cell

The structure is gas-tight and can be used by bubbling an inert gas through the electrolyte to remove or exclude contaminants such as oxygen and water (in an external repository).
Photo-electrochemical H-cells

These are horizontally mounted double chamber photo-electrochemical H-Cells used to study thin film photoanodes and cathodes simultaneously. These units allow for a variety of methods in order to carry out measurements in 2 or 3 electrode settings.
The structure is gas-tight with gas inlets and outlets which can be used to bubble the solution and vent gases from each of the two distinct chambers.
Similarly, and in the case of the photoelectrochemical flow H-Cell, the structure is liquid-tight with electrolyte inlets and outlets, which allow solution flow as well as the emptying of the reaction product from each of the two distinct chambers.
Typical applications include: basic photochemistry, photolysis water splitting, photoinduced charge separation, photocorrosion and CO2 reduction.